Understanding Denmark’s Quality-Driven Healthcare Market and How Electrosurgical Manufacturers Must Align
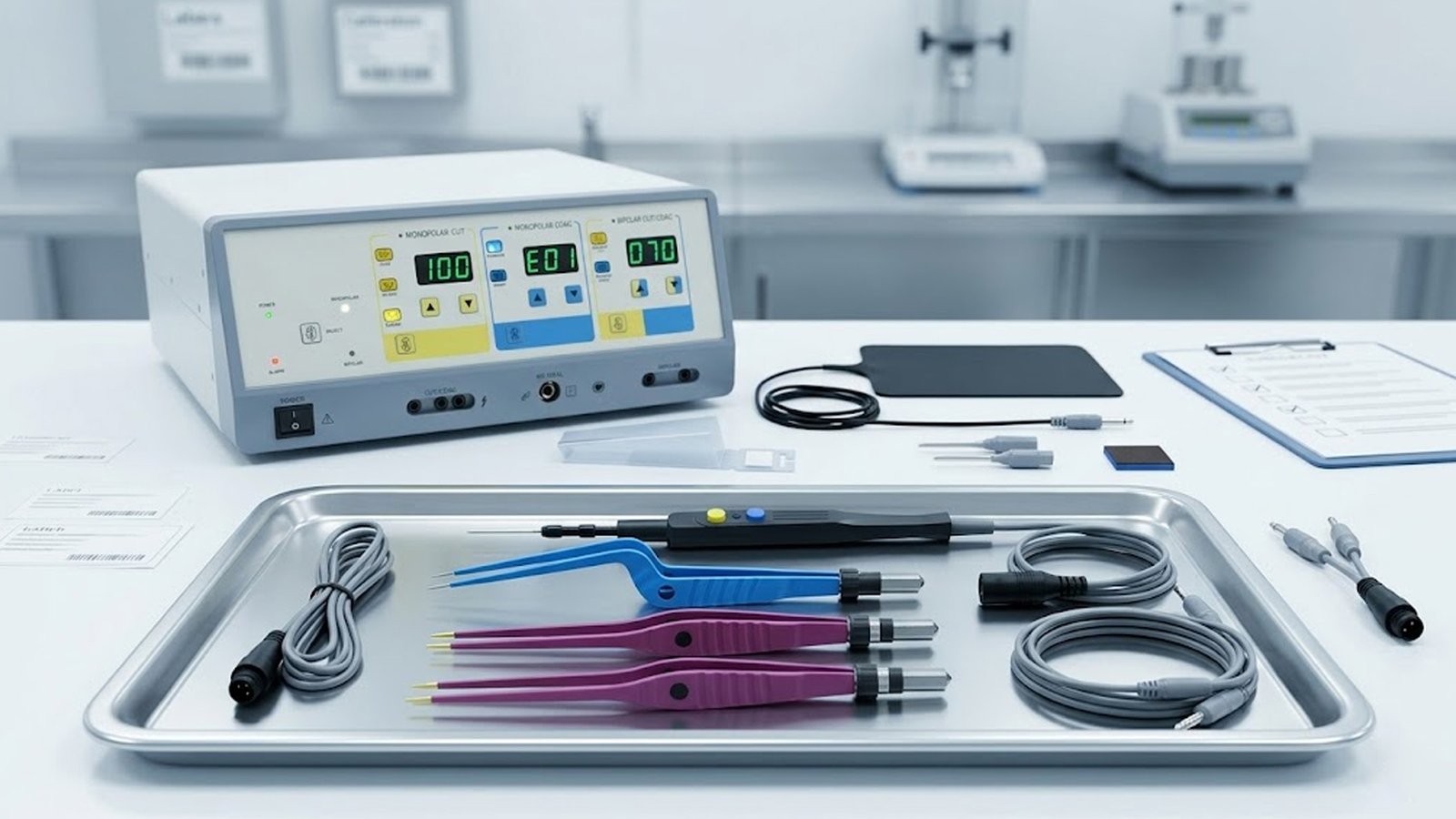
Introduction: Denmark Sets the Benchmark for Surgical Standards
Denmark’s healthcare system is often viewed as a benchmark across Europe — not because it is the largest market, but because it is one of the most disciplined, transparent, and quality-driven.
In Danish operating rooms, electrosurgical instruments are treated as critical clinical tools, not consumable commodities. Every device introduced into a hospital environment is expected to demonstrate consistency, reliability, and long-term safety.
For distributors and OEM partners targeting Denmark in 2026, success depends on understanding one key truth:
Danish hospitals prioritize trust, documentation, and performance continuity above price alone.
How Electrosurgery Is Used Across Danish Operating Rooms
Electrosurgical technology is deeply integrated into Denmark’s surgical workflows. From large university hospitals to regional surgical centers, electrosurgery supports:
- General and colorectal surgery
- Gynecological procedures
- ENT and neurosurgical applications
- Minimally invasive and laparoscopic surgery
Devices such as bipolar forceps, active electrodes, and diathermy machines are evaluated not only on clinical effectiveness, but also on how predictably they perform across repeated procedures.
Danish surgeons expect tools that behave the same way every time — no learning curve surprises, no inconsistent energy delivery.
What Danish Hospitals Expect from Electrosurgical Suppliers
Procurement processes in Denmark are structured and methodical. Decisions are often made by multidisciplinary teams that include clinicians, biomedical engineers, and procurement specialists.
They typically look for:
- Proven electrical safety and stability
- Clear technical specifications
- Consistent manufacturing quality
- Strong documentation support
- Long-term supply reliability
For distributors, this means that representing an electrosurgical brand in Denmark requires confidence — not just in the product, but in the manufacturer behind it.
Bipolar Electrosurgery: A Strong Preference in Denmark
One clear trend in Danish operating rooms is the strong preference for bipolar electrosurgical instruments, especially in procedures where precision and tissue protection are critical.
Bipolar systems are favored because they:
- Limit energy spread to targeted tissue
- Reduce thermal injury risks
- Offer greater control in delicate procedures
As a result, Danish distributors maintain steady demand for bipolar forceps, bipolar electrodes, and compatible connectivity solutions that meet hospital expectations for precision and reliability.
Single-Use vs Reusable Devices in the Danish Context
Denmark maintains a balanced approach between reusable and single-use electrosurgical instruments.
Reusable instruments are commonly used in:
- Public hospitals with established sterilization departments
- Facilities focused on long-term cost efficiency
Single-use instruments are increasingly selected for:
- High-volume procedures
- Infection-sensitive environments
- Workflow-optimized surgical centers
Distributors supplying Denmark often stock both categories, allowing hospitals flexibility based on clinical and operational needs. Manufacturers offering a complete portfolio — including sterile single-use electrosurgical devices — are better positioned for long-term partnerships.
Why Documentation and Traceability Matter More in Denmark
Denmark’s healthcare culture emphasizes accountability. Hospitals expect distributors to provide clear answers to technical and regulatory questions — without delay.
This includes:
- Product identification and traceability
- Consistent labeling and packaging
- Clear instructions for use
- Stable product specifications across batches
Manufacturers who maintain disciplined documentation practices make it easier for distributors to operate confidently in audits, tenders, and long-term supply agreements.
Distributor–Manufacturer Relationships in Denmark
In Denmark, transactional supply relationships rarely last. Hospitals prefer continuity, and distributors reflect this preference when choosing manufacturing partners.
Distributors value manufacturers who:
- Communicate transparently
- Maintain stable production timelines
- Support OEM and private-label programs
- Understand European hospital expectations
A manufacturer that aligns with these values becomes part of the distributor’s long-term strategy — not just their catalog.
Electro Range MFG Co. and the Danish Market
Electro Range MFG Co. (ERMC) manufactures electrosurgical instruments with an understanding of markets like Denmark, where consistency and trust define purchasing decisions.
By offering:
- Electrosurgical forceps
- Bipolar and monopolar electrodes
- Diathermy systems
- Laparoscopic electrosurgical instruments
- Reusable and single-use options
ERMC enables Danish distributors to build focused, reliable product portfolios that meet both clinical and regulatory expectations.
Why Denmark Matters in Europe’s 2026 Electrosurgical Landscape
Denmark may not be the largest European market, but it is one of the most influential.
Success in Denmark signals:
- Strong product credibility
- High compliance standards
- Long-term distributor trust
For manufacturers and distributors alike, Denmark represents a market where quality speaks louder than marketing.
Let’s Support Denmark’s Surgical Ecosystem Together
If you are a Danish medical device distributor, procurement specialist, or OEM partner seeking dependable electrosurgical manufacturing support, we welcome a professional discussion.
📞 Call us: +92 321 610 0080
🌐 Website: https://electrorange.com/
📩 Distributor & OEM inquiries welcome

