Understanding the Differences That Matter to Surgeons, Distributors, and Procurement Teams Across Europe
Introduction: Why the Bipolar vs Monopolar Question Still Matters in Europe
Across European operating rooms, electrosurgery is a daily clinical reality. Yet one question continues to shape purchasing decisions, product portfolios, and distributor strategies: bipolar or monopolar electrosurgery?
For surgeons, the answer often depends on precision, tissue sensitivity, and procedural control. For distributors and procurement teams in Denmark, Sweden, Norway, Portugal, Greece, and Poland, the answer carries an additional layer — commercial viability, compliance, and long-term demand stability.
Understanding the practical differences between bipolar and monopolar electrosurgery is essential not only for clinical alignment, but also for building a balanced, future-ready electrosurgical portfolio in Europe.
Monopolar Electrosurgery: Versatility and Broad Clinical Use
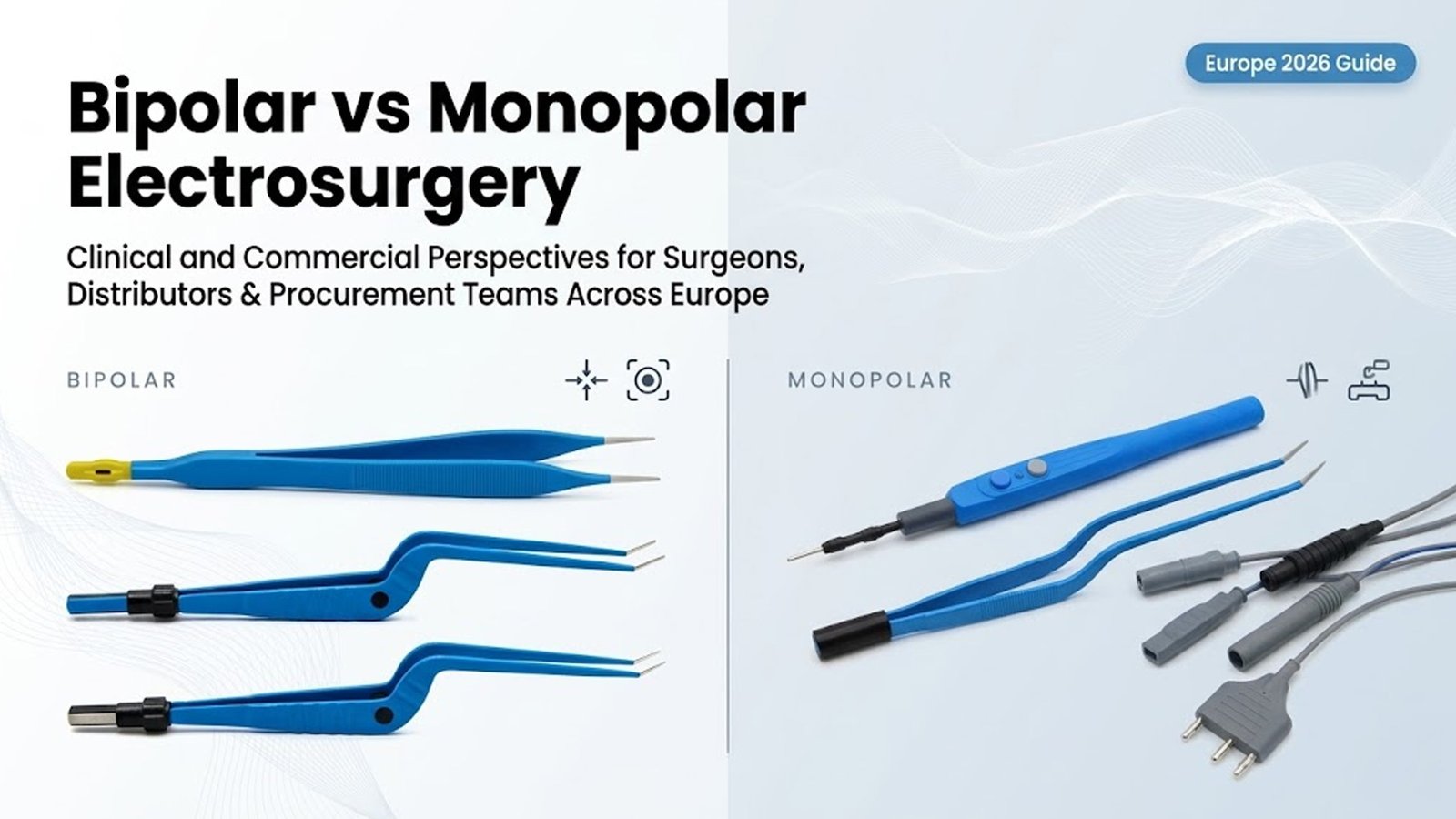
Monopolar electrosurgery has long been a foundation of surgical practice across Europe. In a monopolar system, electrical current flows from the active electrode, through the patient’s body, and returns via a neutral electrode plate.
This setup makes monopolar instruments especially useful for:
- General surgery
- Open surgical procedures
- Procedures requiring wider tissue effect
- Applications where speed and reach are priorities
From a distributor perspective, monopolar instruments remain essential. Products such as active monopolar electrodes, compatible cables, and patient plates continue to see consistent demand across European hospitals.
Monopolar electrosurgery is often favored in settings where:
- Surgical teams are well-trained in its use
- Procedure variety is high
- Cost efficiency is a consideration
This explains why monopolar systems remain deeply embedded in Europe’s surgical infrastructure.
Bipolar Electrosurgery: Precision, Control, and Targeted Energy
Bipolar electrosurgery represents a more targeted approach. In bipolar systems, electrical current flows only between the two tips of the instrument, passing directly through the tissue being treated.
This design significantly reduces:
- Unintended thermal spread
- Risk to surrounding tissue
- Dependence on patient return electrodes
As a result, bipolar electrosurgery is widely preferred for:
- Neurosurgery
- ENT procedures
- Gynecological surgery
- Pediatric and delicate tissue applications
European hospitals increasingly specify bipolar solutions in tenders, driving steady demand for products such as bipolar forceps and dedicated bipolar electrodes.
From a commercial standpoint, bipolar instruments are often associated with:
- Higher perceived safety
- Stronger clinical justification
- Growing preference in regulated environments
Clinical Decision-Making: When Bipolar or Monopolar Is Preferred
In practice, European surgeons rarely view bipolar and monopolar electrosurgery as competing technologies. Instead, they see them as complementary tools.
Monopolar electrosurgery is typically chosen when:
- Broader tissue effect is required
- Procedure speed is critical
- Access and reach are priorities
Bipolar electrosurgery is preferred when:
- Precision is essential
- Thermal spread must be minimized
- Work is performed near sensitive structures
For distributors and procurement teams, this reinforces a key point: a well-balanced portfolio must include both technologies.
Commercial Implications for European Distributors
Across Europe, hospitals increasingly expect distributors to offer flexible electrosurgical solutions rather than single-technology portfolios.
From a commercial perspective:
- Monopolar products ensure volume stability
- Bipolar products support premium positioning
- Single-use options align with infection-control priorities
Distributors supplying both reusable and sterile single-use electrosurgical instruments are better positioned to meet diverse hospital requirements across different European healthcare systems.
Manufacturers who support this portfolio diversity enable distributors to respond effectively to tenders, framework agreements, and evolving clinical preferences.
Energy Control and Equipment Compatibility
Both bipolar and monopolar instruments depend heavily on stable energy sources and reliable connectivity.
In Europe, compatibility between:
- Electrosurgical instruments
- Energy generators
- Cables and connectors
is a major evaluation factor. Products such as electrosurgical cables and connectors play a critical role in ensuring consistent performance, regardless of whether bipolar or monopolar systems are used.
Distributors increasingly favor manufacturers who understand system-level compatibility, not just individual instruments.
Regulatory and Safety Expectations in Europe
European healthcare markets apply the same regulatory rigor to both bipolar and monopolar electrosurgical devices.
Procurement teams expect:
- Clear product identification
- Consistent electrical safety performance
- Transparent documentation
- Alignment with European medical device requirements
This reinforces the importance of working with manufacturers who maintain disciplined production processes and regulatory awareness across their entire electrosurgical range.
Electro Range MFG Co.: Supporting Both Bipolar and Monopolar Needs
Electro Range MFG Co. (ERMC) manufactures a comprehensive range of electrosurgical instruments designed to support both bipolar and monopolar applications.
By offering:
- Reusable and single-use bipolar forceps
- Active monopolar electrodes
- Compatible cables and accessories
- Electrosurgical systems aligned with European expectations
ERMC enables distributors and OEM partners to build balanced, compliant portfolios suited for diverse European clinical environments.
Why Understanding This Difference Matters in 2026
As Europe continues to modernize surgical care, the demand for precision, safety, and flexibility will only increase.
Distributors who understand how bipolar and monopolar electrosurgery fit into real clinical workflows are better positioned to support hospitals — and to grow sustainably in competitive markets.
Let’s Align Your Electrosurgical Portfolio for Europe
If you are a medical device distributor, procurement professional, or OEM partner seeking to strengthen your electrosurgical offering across Europe, we are ready to support an informed, transparent discussion.
📞 Call us: +92 321 610 0080
🌐 Website: https://electrorange.com/
📩 Distributor & OEM inquiries welcome